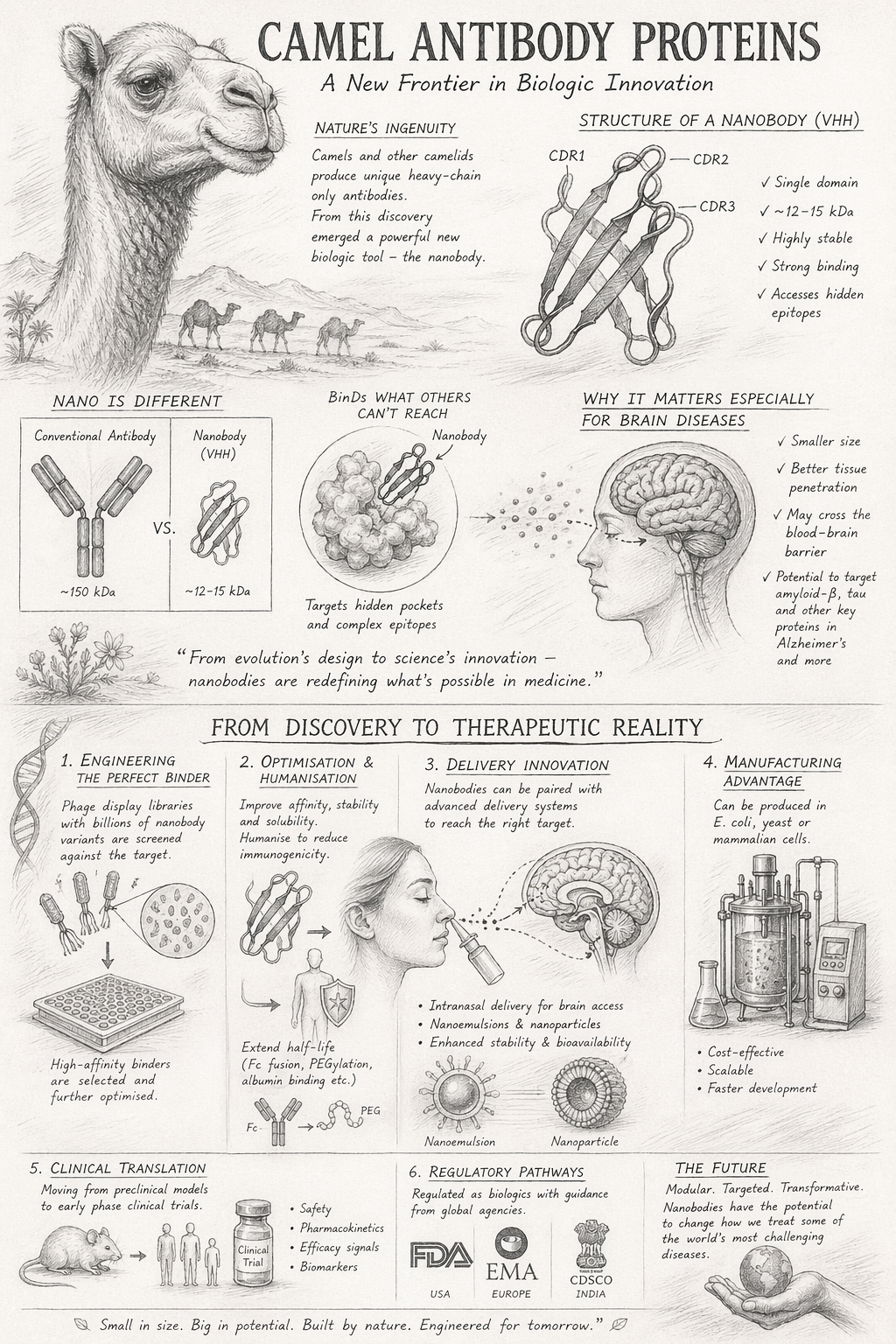
Extracellular Matrix:
The Hidden Architect of Life and Therapeutics
From structural scaffold to biological command centre — understanding the ECM and its emerging role in next-generation medicine.
Section 1 — Understanding the ECM
The overlooked system that defines life between cells
When discussing biology, the focus traditionally centres on cells, genes, and proteins. Yet, equally fundamental — though historically underappreciated — is the extracellular matrix (ECM): a highly organised, dynamic network that occupies the space between cells and orchestrates tissue function at multiple levels.
Far from being an inert filler, the ECM is now recognised as a critical regulator of biological systems, shaping how cells behave, communicate, and respond to their environment. Contemporary research positions the ECM not merely as structural support, but as a central control interface for tissue homeostasis, repair, and disease progression.
"The ECM is not a passive backdrop to cellular life — it is an active, signal-rich environment that instructs, shapes, and sustains the tissues it pervades."
What exactly is the extracellular matrix?
At its core, the ECM is a three-dimensional macromolecular network. Together, its components form a biochemical and biomechanical framework that surrounds and supports cells within tissues.
Structural Proteins
Collagen and elastin provide tensile strength, resilience, and mechanical integrity to tissues.
Adhesive Glycoproteins
Fibronectin and laminins mediate cell attachment, migration, and tissue-specific organisation.
Proteoglycans
Bind growth factors, regulate water retention, and modulate signalling within the matrix.
Glycosaminoglycans (GAGs)
Long polysaccharide chains that contribute to the hydrated gel-like properties of the ECM.
However, this definition only scratches the surface. The ECM is dynamic — constantly synthesised, remodelled, and degraded; tissue-specific — with composition varying across organs; and signal-rich — integrating both mechanical and biochemical cues.
Structural organisation: More than just a scaffold
The ECM is broadly classified into two major compartments:
Interstitial Matrix
Found between cells in connective tissues. Rich in fibrillar collagens, elastin, and proteoglycans — providing tensile strength and mechanical resilience.
Basement Membrane
A thin, specialised layer underlying epithelial and endothelial cells. Composed mainly of type IV collagen and laminins — acting as both structural support and selective barrier.
This dual architecture enables the ECM to simultaneously maintain tissue architecture, regulate cell polarity and organisation, and serve as a biological interface between tissue compartments.
The ECM as a signalling powerhouse
One of the most transformative insights in modern biology is that the ECM is not passive — it is biologically instructive. It regulates key cellular processes including adhesion, migration, proliferation, differentiation, and apoptosis through two major mechanisms:
Biochemical Signalling
ECM molecules bind to cell surface receptors (e.g. integrins), triggering intracellular pathways that influence gene expression and cellular behaviour.
Mechanotransduction
Cells sense physical properties of the ECM — stiffness, elasticity, topography — directly influencing cell fate decisions, stem cell differentiation, and disease progression.
Cells migrate preferentially towards stiffer environments — a process known as durotaxis — and ECM stiffness can alter gene expression patterns linked to cancer and fibrosis.
A dynamic system: Continuous remodelling
Unlike static materials, the ECM is in a constant state of flux. It undergoes deposition by cells (e.g. fibroblasts), degradation by enzymes (e.g. matrix metalloproteinases), and reorganisation in response to environmental cues. This process — known as ECM remodelling — is essential for embryonic development, wound healing, and tissue regeneration.
However, when dysregulated, it becomes a primary driver of disease.
ECM in disease: From consequence to cause
Emerging evidence now positions ECM dysfunction not merely as a by-product of disease, but as a primary driver of pathology.
Excess ECM deposition leads to progressive tissue stiffening and impaired organ function — affecting the liver, lung, and heart.
Altered ECM architecture promotes tumour invasion, immune evasion, and resistance to therapeutics by reshaping the tumour microenvironment.
ECM degradation products — known as matrikines — can act as signalling molecules that influence immune responses and disease progression in neurological conditions.
In many chronic conditions, a self-reinforcing loop emerges:
This feedback loop is now considered a central axis in diseases ranging from cancer to cardiovascular disorders.
ECM as the blueprint for regenerative medicine
The realisation that ECM governs cellular behaviour has fundamentally shifted biomedical research. Instead of asking, "How do we replace damaged tissue?", the field is now asking, "How do we recreate the microenvironment that enables tissues to heal themselves?"
This has led to the rise of ECM-inspired and ECM-derived biomaterials — including decellularised ECM scaffolds, ECM hydrogels, and bioengineered matrices — designed to replicate the ECM's structural complexity, biochemical signalling, and mechanical properties.
The ECM market is expanding rapidly, driven by rising chronic disease burden, demand for advanced biomaterials, and advances in 3D cell culture and bioprinting. Applications span regenerative medicine, wound healing, drug delivery, and oncology.
Section 2 — From Insight to Therapeutic Innovation
Translating ECM science into next-generation therapies and future opportunities
The growing understanding of the ECM has catalysed a fundamental shift in medicine: from targeting isolated molecular pathways to engineering the cellular microenvironment itself. Where traditional therapeutics aim to modulate receptors or enzymes, ECM-based approaches seek to reprogramme tissue behaviour at a systems level — restoring the conditions under which cells can function, repair, and regenerate appropriately.
"Microenvironment-driven therapies sit at the intersection of biologics, devices, and advanced biomaterials — and they work with biology, rather than overriding it."
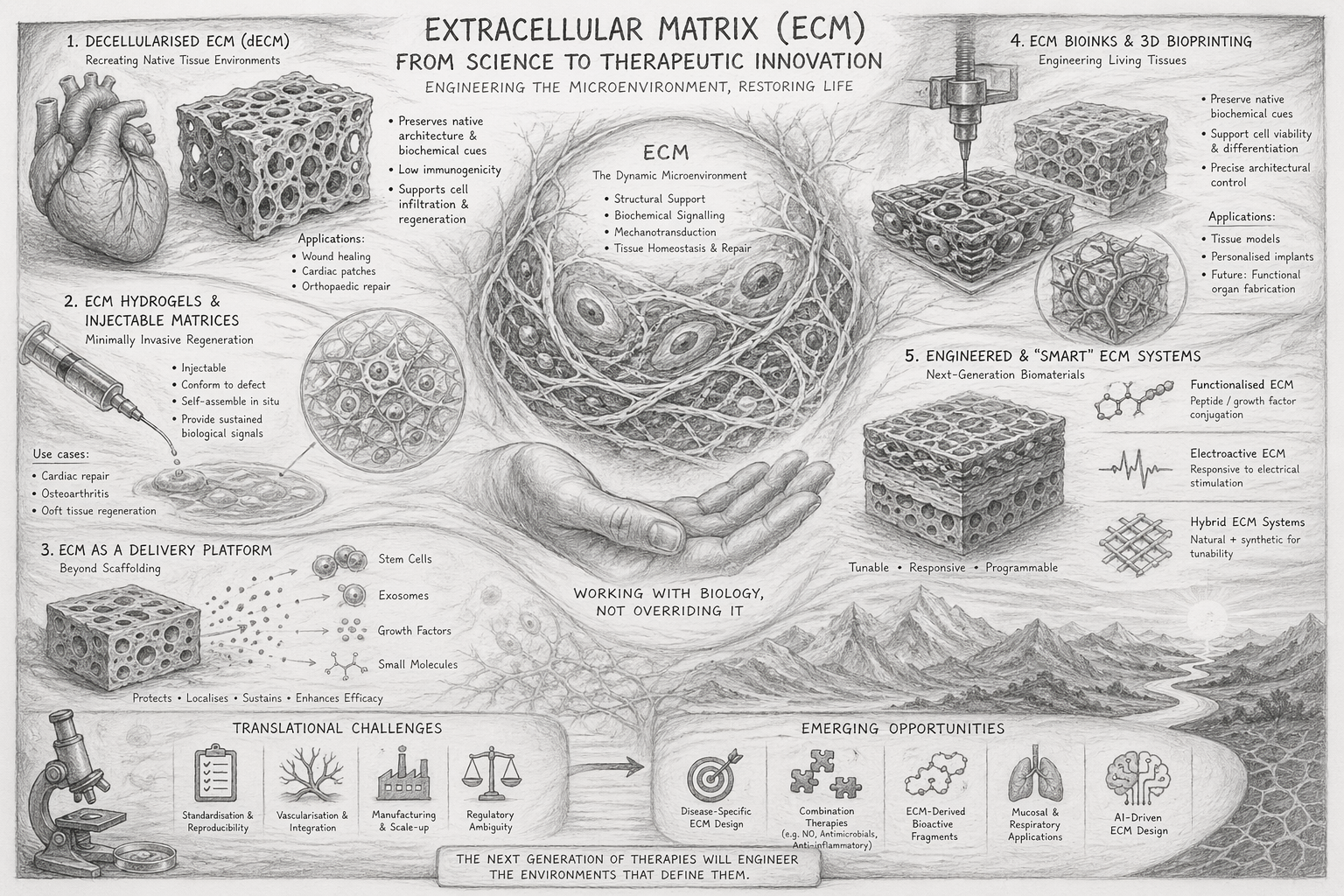
The extracellular matrix — a three-dimensional network of structural proteins, glycoproteins, and proteoglycans that orchestrates tissue function and repair.
Decellularised ECM — Recreating Native Tissue Environments
Decellularised ECM (dECM) involves removing cellular components from tissues whilst preserving the native extracellular architecture. The resulting biologically active scaffold retains structural proteins, growth factors, and tissue-specific signalling cues — mimicking the native microenvironment with high fidelity, reducing immunogenicity, and supporting cell infiltration, angiogenesis, and regeneration.
- Dermal substitutes for burns and chronic wounds
- Cardiac patches post-myocardial infarction
- Orthopaedic and musculoskeletal repair
The next phase: standardised, reproducible dECM systems that overcome batch-to-batch variability and scalability limitations.
ECM Hydrogels & Injectable Matrices — Minimally Invasive Regeneration
ECM-derived hydrogels represent a major advancement in delivering regenerative therapies without surgery. These injectable materials conform to tissue defects, self-assemble into 3D networks in situ, and provide sustained biochemical signalling — demonstrating improvements in tissue perfusion, functional recovery, and reduced fibrosis.
- Cardiac repair following infarction
- Osteoarthritis and cartilage regeneration
- Soft tissue reconstruction
ECM as a Delivery Platform — Beyond Scaffolding
Rather than acting as a passive carrier, the ECM can bind and protect bioactive molecules, release them in a controlled and context-dependent manner, and enhance localisation and retention. This positions ECM as a biologically intelligent delivery system — with synergistic effects between the matrix's own signalling and therapeutic payloads such as stem cells, exosomes, growth factors, and small molecules.
ECM Bioinks & 3D Bioprinting — Engineering Living Tissues
The integration of ECM with 3D bioprinting has unlocked the ability to fabricate complex, spatially organised tissue constructs. ECM-based bioinks preserve native biochemical cues, support cell viability and differentiation, and enable precise architectural control — with advances including patient-specific bioinks, multi-material printing for vascularised constructs, and AI-guided scaffold design.
- Tissue models for drug testing
- Personalised implants
- Long-term goal: functional organ fabrication
Engineered & "Smart" ECM Systems — Programmable Biomaterials
Next-generation ECM systems are being engineered to overcome the limitations of native materials. These include functionalised ECM conjugated with peptides or growth factors, electroactive ECM responsive to electrical stimulation, and hybrid systems combining natural and synthetic materials for tunability — enabling controlled degradation rates, tunable mechanical properties, and enhanced signalling precision.
Translational challenges: Where innovation is still needed
Despite rapid progress, ECM-based therapies face several critical bottlenecks — each representing a strategic entry point for innovation:
Variability in source tissues and processing methods, combined with a lack of uniform quality benchmarks, limits clinical translation.
Limited blood vessel formation in engineered tissues and poor long-term viability in large constructs remain key hurdles.
Complex processing workflows and difficulty achieving industrial-scale production constrain commercial viability.
ECM products sit between biologics, medical devices, and combination products — creating uncertainty in regulatory pathways across jurisdictions.
Emerging opportunities for research and development
The ECM field remains far from saturated. Several high-value directions are gaining traction:
Tailoring ECM composition for cardiac repair, neurodegenerative conditions, and chronic wounds.
Integrating ECM with gasotransmitters (e.g. nitric oxide), antimicrobial, and anti-inflammatory agents.
ECM-derived bioactive fragments offering defined signalling functions with greater regulatory clarity.
Nasal and pulmonary delivery systems for barrier restoration and regenerative therapies.
Predictive modelling of ECM composition and optimisation for specific tissues and disease contexts.
A New Therapeutic Paradigm
The extracellular matrix is rapidly transitioning from a supporting actor to a central therapeutic platform. The next generation of therapies will not simply target cells — they will engineer the environments that define them. For organisations positioned at the intersection of formulation science, biomaterials, and translational medicine, the ECM represents not just an opportunity, but a strategic frontier.