Predictive Intelligence for Development Decisions
A proprietary platform that integrates scientific evidence, predicts human pharmacokinetics, and defines formulation and development decisions
Intelligence
From molecule selection to clinical positioning — guided by predictive evidence, not trial-and-error.
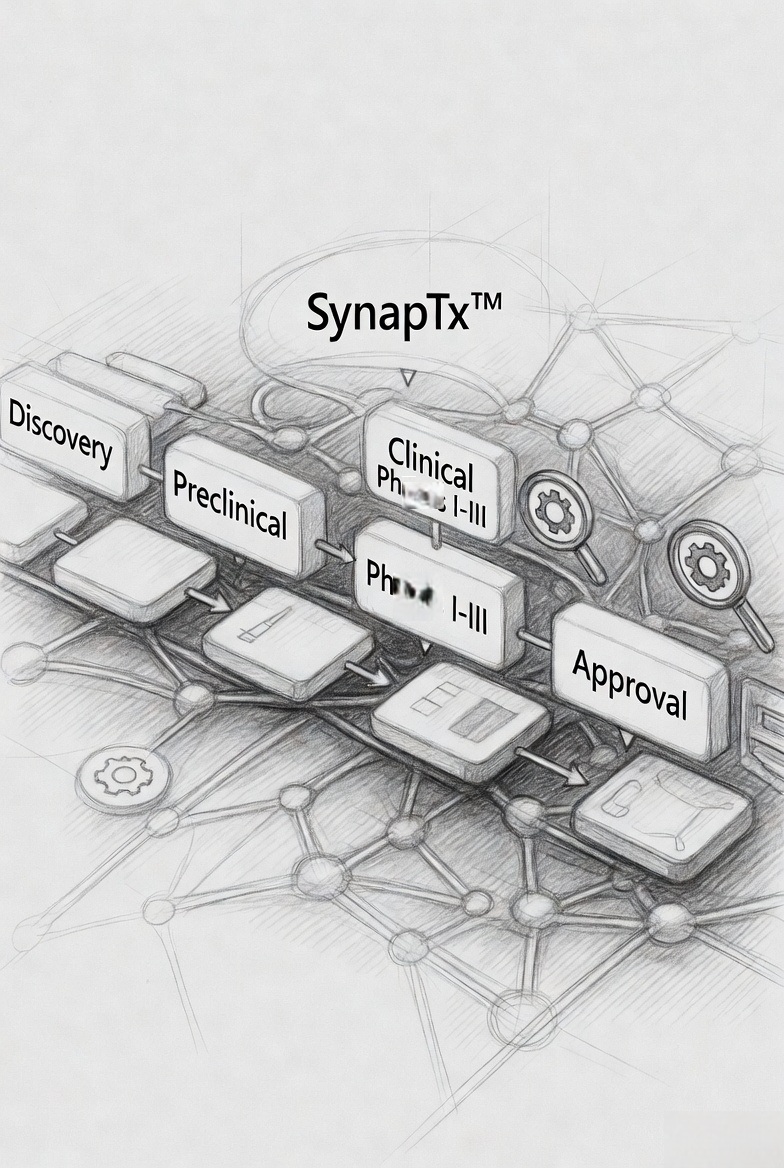
SynapTx™ functions as a unified decision engine across the drug development lifecycle — integrating scientific literature, pharmacokinetic modelling, formulation datasets, and global development data enabling teams to evaluate molecular potential, define formulation strategies, and engineer development pathways.
It enables prediction of human exposure and identification of formulation and CMC constraints before physical development begins.
The problem
01
Drug development remains fundamentally unpredictable
Despite advances in molecular discovery, the ability to reliably predict human pharmacokinetics and clinical performance remains limited. As a result, development programmes are often driven by trial-and-error rather than informed foresight.
This leads to:
- Late-stage failures after significant investment
- Iterative, trial-and-error formulation cycles
- Delays in clinical progression
- Underutilisation of existing scientific, clinical, and formulation knowledge
INTRODUCING SYNAPTX
02

SynapTx™
A unified intelligence layer for drug development
SynapTx™ is a proprietary, IP-secure platform designed to bring predictability to development decisions
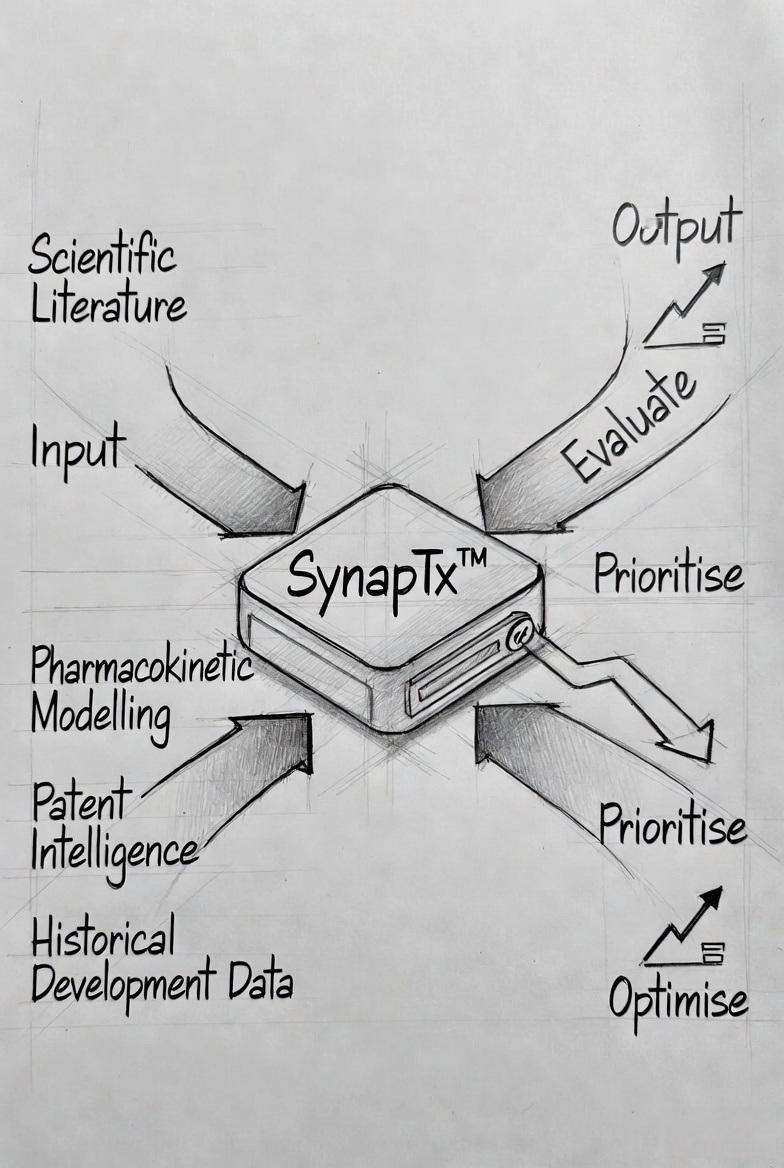
It integrates scientific literature, pharmacokinetic modelling, patent intelligence, and historical development data into a single decision engine — enabling teams to evaluate, prioritise, and optimise development strategies early.
It continuously analyses:
- Global scientific and clinical literature
- Pharmacokinetic and formulation datasets
- Patent landscapes and competitive intelligence
- Historical development successes and failures
- Degradation kinetics, excipient compatibility, and manufacturability constraints
Transforming fragmented data into structured, decision-ready insight
SynapTx™ functions as the central intelligence engine across the development lifecycle.
From data aggregation to actionable strategy
03
Core intelligence capabilities powering SynapTx™
Scientific Evidence Integration
Aggregates and interprets large-scale scientific and clinical literature to identify mechanisms, development precedents, and formulation-relevant insights
Predictive Pharmacokinetic Modelling
Simulates human pharmacokinetics and exposure to inform formulation design, dosing strategy, and clinical positioning
Patent & Landscape Intelligence
Analyses global patent filings and competitive landscapes to identify white spaces, development risks, and differentiation opportunities.
Repurposing & Opportunity Mapping
Identifies alternative therapeutic applications and development pathways based on integrated mechanistic, clinical, and formulation insights
HOW IT IMPACTS DEVELOPMENT
04
From fragmented data to execution-ready decisions
By integrating scientific evidence, predictive modelling, and development intelligence, SynapTx™ enables more informed, efficient, and strategically aligned decision-making across the development lifecycle.
SynapTx™ enables:
- Early identification of viable and executable development pathways
- Reduction in iterative formulation cycles through predictive design
- Improved predictability of human exposure and clinical outcomes
- Alignment with regulatory expectations from early development
- Accelerated progression from molecule to clinic
Identification
Accelerate
Predictability
Neural
Increase response
Easily integrate
Identify
Localised
Generative
Agile
Precise
Rigorous
FROM INSIGHT TO EXECUTION
05
Insight is not theoretical—it drives real-world development decisions
SynapTx™ is embedded directly within development workflows, ensuring that insights are directly translated into actionable strategies across formulation, clinical planning, and regulatory execution.
It does not operate as a standalone analytical tool—it functions as a decision engine that defines and shapes each stage of development
SynapTx™ directly informs:
- Molecular engineering and formulation design
- Human pharmacokinetic prediction and exposure modelling
- Translational development and clinical strategy
- CMC strategy, manufacturability, and regulatory planning
SECURE BY DESIGN
06

Secure
Designed for secure, IP-protected drug development
SynapTx™ operates within secure, controlled environments—ensuring full protection of proprietary data, development strategies, and intellectual property
The platform is designed to support confidential internal programmes as well as strategic collaborations, without compromising data integrity or ownership.
Built for confidentiality and IP integrity
- Locally hosted infrastructure
- No external data exposure
- Full control over proprietary datasets
- Designed for secure collaboration
WHY SYNAPTX™ IS DIFFERENT
07
A fundamentally different approach to drug development intelligence
SynapTx™ is not a standalone analytics tool. It is built as an integrated intelligence system that connects prediction, strategy, and execution across the drug development lifecycle.
By integrating scientific, clinical, formulation, regulatory, and competitive intelligence within a single framework, it enables more cohesive and confident development decisions.
Unlike conventional modelling platforms, SynapTx incorporates formulation behaviour, degradation kinetics, and CMC constraints—enabling prediction of not only biological response, but manufacturable and clinically viable solutions.
Differentiation Points
- Purpose-built for pharmaceutical development
- Integrates scientific, regulatory, and commercial intelligence in one system
- Connects predictive insight directly to formulation and development strategy
- Enables earlier, more confident decision-making across programmes
- Designed to support both internal pipelines and strategic partnerships
Bring predictability to your development decisions
Whether you are evaluating a new molecule, addressing formulation challenges, or preparing for clinical translation, SynapTx™ enables earlier, more informed, and lower-risk development decisions.
Lets talk
Confidential discussions. IP-safe engagement. Scientific-first approach.