The Hidden Bottleneck in Pharma:
Why CMC and Formulation Decide Clinical Success
Discovery biology takes centre stage — but the true crucible of drug development is Chemistry, Manufacturing, and Controls. Here is why formulation science is now the ultimate competitive advantage.
Introduction
The real science happens
after discovery
In the theatre of drug development, discovery biology and high-throughput screening typically take centre stage. The industry rightfully celebrates the identification of novel targets and the synthesis of molecules with exquisite nanomolar affinity. However, the reality of translational medicine is far more unforgiving.
The true crucible — where promising early-stage assets either evolve into life-saving therapies or become costly statistics — is Chemistry, Manufacturing, and Controls (CMC).
“For decades, CMC was viewed as a backend, operational necessity: a box to tick after the ‘real’ science of discovery was complete. Today, that paradigm has entirely shifted.”
The escalating complexity of modern therapeutics dictates that formulation and manufacturability are no longer afterthoughts; they are the fundamental determinants of clinical and commercial viability.
The Developability Challenge
The ‘Developability’ Crisis and the
Demise of the Rule of Five
The primary driver behind the rising prominence of CMC is the evolving nature of the Active Pharmaceutical Ingredient (API) itself. Historically, drug discovery adhered closely to Lipinski’s Rule of Five, selecting for small molecules that were inherently water-soluble and easily absorbed.
However, as we pursue more complex biological targets — such as manipulating TLR5 signalling in the tumour microenvironment or designing highly specific kinase inhibitors — we are generating molecules that are decidedly non-compliant. The contemporary oncology and immunology pipelines are heavily populated by highly lipophilic, high-molecular-weight compounds.
Advanced formulation science bridges the gap between molecular discovery and clinically viable therapeutics — transforming ‘brick dust’ molecules into developable assets.
These molecules, often categorised as Biopharmaceutics Classification System (BCS) Class II or IV, are notoriously insoluble. They are colloquially referred to as ‘brick dust’ or ‘grease’. An asset can demonstrate perfect target engagement in a controlled in vitro assay, but if its physicochemical properties prevent it from dissolving in the gastrointestinal tract or remaining stable in human plasma, its theoretical efficacy is clinically useless.
This ‘developability crisis’ means that without early and aggressive formulation intervention, promising molecules will fail in Phase I due to catastrophic pharmacokinetic shortfalls — not because the pharmacology was wrong, but because the delivery was never engineered.
Advanced Formulation Science
Formulation as a
Pharmacokinetic Bridge
To overcome these physiological hurdles, formulation science has evolved from basic compounding into sophisticated biophysical engineering. The formulation is now an active partner in defining the drug’s pharmacokinetic profile, enabling developers to manipulate systemic exposure, bypass first-pass metabolism, and mitigate dose-limiting toxicities.
Several advanced CMC strategies are currently at the forefront of rescuing complex APIs:
Amorphous Solid Dispersions (ASDs)
For the most intractable ‘brick dust’ molecules, simply milling the API is insufficient. By using hot-melt extrusion or spray drying to trap the drug in an amorphous, high-energy state within a polymer matrix, formulators can force a supersaturated state in the gut, drastically enhancing oral bioavailability.
Complex Architectural Dosage Forms
When systemic toxicities are tied to peak plasma concentrations (Cmax), standard immediate-release capsules are inadequate. The engineering of sophisticated delayed-release and bilayer tablets allows for an immediate loading dose followed by a precisely controlled sustained-release matrix, maintaining the API within a narrow therapeutic window for extended periods.
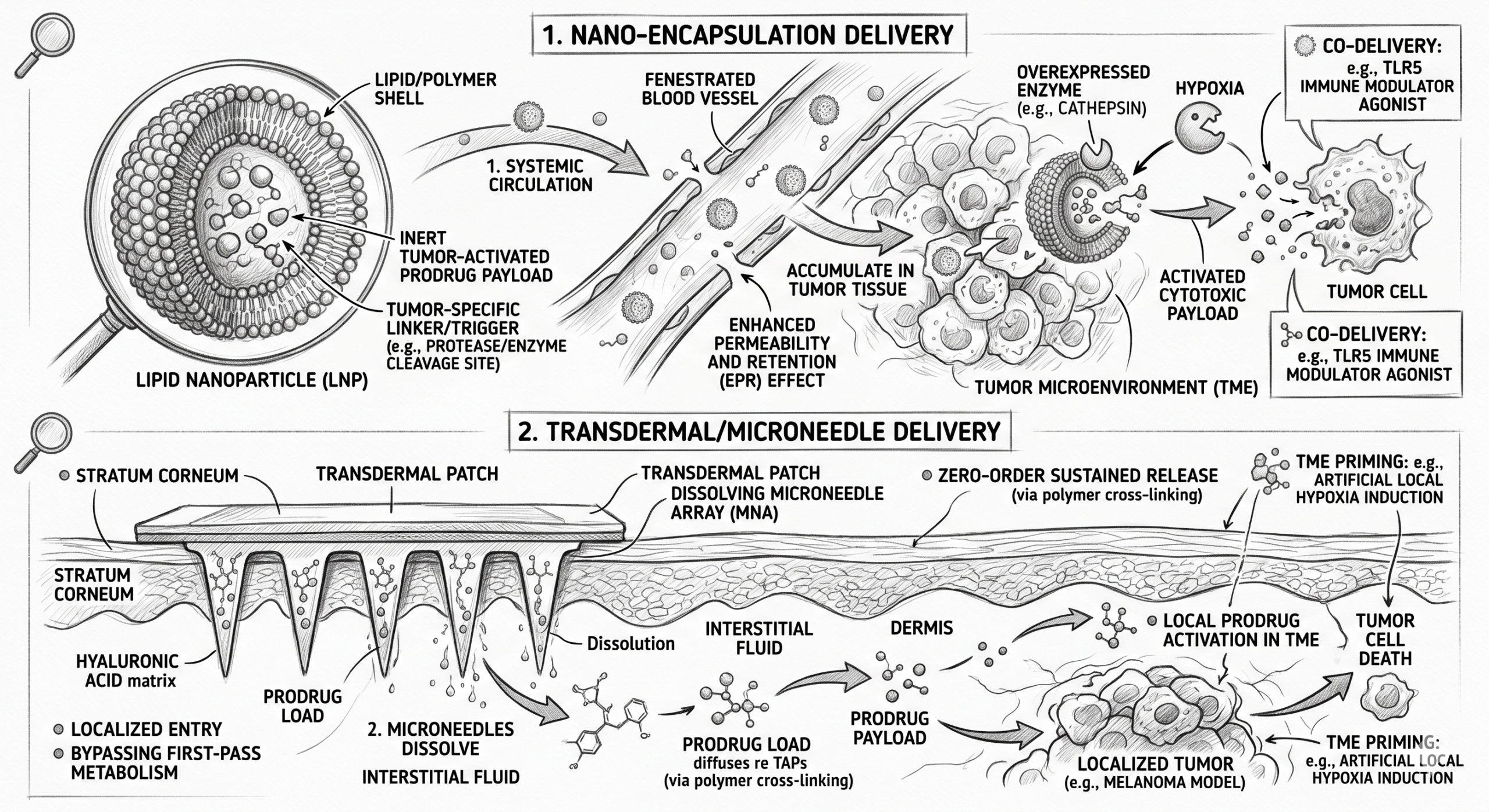
Nanomedicine and Peptide Delivery
As the industry leans into modalities such as oral peptides and complex small-molecule targeted therapies, the formulation must act as a shield. Nano-encapsulation techniques are critical for protecting fragile payloads from the harsh enzymatic environment of the stomach, ensuring they survive long enough to cross the intestinal epithelium or accumulate in highly vascularised tumour beds via the Enhanced Permeability and Retention (EPR) effect.
Manufacturing Challenges
The Scale-Up Chasm:
From Bench to Bioreactor
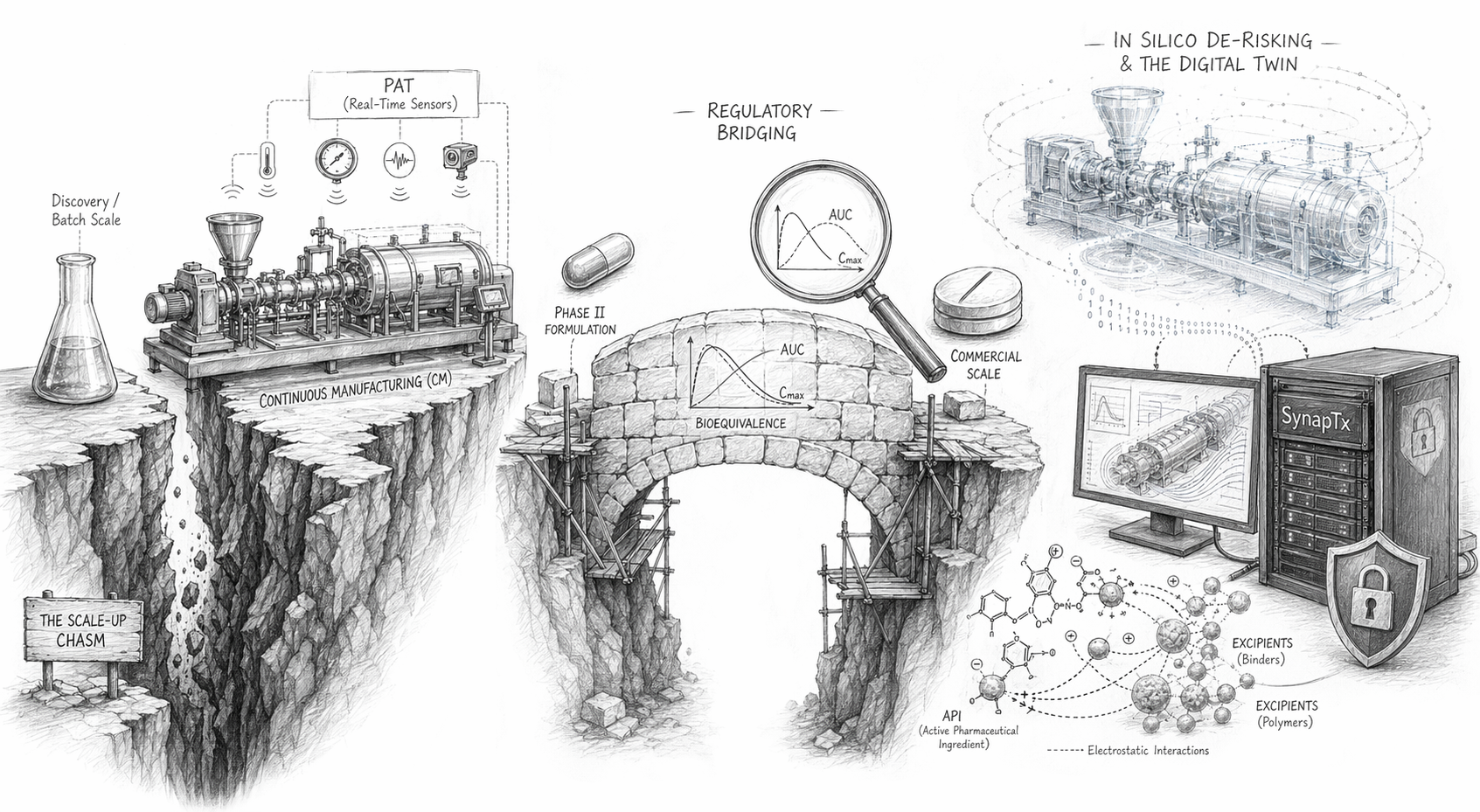
Beyond the biophysics of formulation, the ‘C’ and ‘M’ in CMC represent an equally formidable barrier: Chemistry and Manufacturing. The synthesis of a novel small molecule in a research laboratory is typically optimised for speed and yield, often requiring hazardous reagents, extreme temperatures, or complex chromatographic purifications to produce a few milligrams for mouse xenograft models.
However, the leap to clinical trials requires kilograms of API manufactured under strict current Good Manufacturing Practice (cGMP) conditions. The jump from small-scale synthesis to commercial production is where many promising therapies meet their demise.
“The inability to consistently reproduce a stable polymorph of the API — where a sudden shift in crystal structure during scale-up completely alters the drug’s solubility and safety profile — has derailed countless late-stage clinical programmes.”
If a complex, multi-step enantioselective synthesis cannot be streamlined, made environmentally sustainable, and rendered economically viable at scale, the asset is dead on arrival. To navigate this, the industry is undergoing a profound paradigm shift in how drugs are manufactured and regulated.
Manufacturing Innovation
Continuous Manufacturing:
The End of the Batch Paradigm
For over a century, pharmaceutical manufacturing relied on rigid batch processing. An API would be synthesised in one facility, shipped to another, milled, mixed with excipients in massive vats, and finally pressed into tablets. Each step required stopping, testing, and waiting. This fragmented approach is inherently inefficient and highly susceptible to batch-to-batch variability.
The modern solution, strongly advocated by global regulatory bodies including the FDA and EMA, is Continuous Manufacturing (CM). In a CM facility, raw materials are fed into one end of a continuous, enclosed system, and finished dosage forms emerge from the other in a ceaseless flow.
Key enablers of continuous manufacturing
Process Analytical Technology (PAT)
The backbone of CM. Rather than testing a subset of finished tablets, PAT utilises in-line sensors — such as near-infrared spectroscopy — to monitor chemical composition, moisture content, and particle size in real-time, hundreds of times per second.
Real-Time Release Testing
Because quality is continuously verified throughout the process, out-of-specification material can be automatically diverted without scrapping an entire batch. This level of rigorous control frequently allows for real-time release, bypassing the lengthy quarantine periods associated with traditional quality control.
Regulatory Landscape
Regulatory Intricacies:
The Cost of CMC Variations
The regulatory landscape for CMC is governed by stringent International Council for Harmonisation (ICH) guidelines — specifically Q8, Q9, Q10, and Q11 — which emphasise Quality by Design (QbD). QbD mandates that quality cannot simply be tested into a product; it must be designed into it from the outset.
ICH Guidelines governing CMC
These guidelines collectively enforce that quality must be engineered into a product from inception — not validated after the fact. Sponsors who fail to establish a robust CMC strategy early will face severe regulatory and commercial consequences.
The penalty for failing to establish a robust CMC strategy early is severe. If a sponsor needs to alter their formulation or manufacturing process mid-way through Phase III — for instance, changing from an early-stage capsule to a commercial-grade bilayer tablet — regulators will demand a Bridging Study.
A bridging study is an expensive, time-consuming clinical trial required to prove that the new formulation exhibits bioequivalence to the old one. If the new commercial formulation alters the Cmax or Area Under the Curve (AUC) beyond an incredibly tight statistical margin, the entire clinical dataset generated by the old formulation may be invalidated. This administrative catastrophe can delay a drug’s time-to-market by years, allowing competitors to capture the space.
Predictive Development
In Silico Modelling:
De-risking Formulation Early
The most effective way to avoid late-stage CMC failures and bridging studies is to predict them before physical manufacturing ever begins. The integration of predictive computational modelling has become the new gold standard for formulation development.
Rather than relying entirely on empirical trial and error, modern formulation scientists and biostatisticians leverage secure, localised AI platforms — such as SynapTx — to run complex thermodynamic and kinetic simulations. These high-parameter, in-house engines can securely process proprietary molecular structures against vast datasets to achieve the following:
Predict Excipient Compatibility
Algorithms can map the electrostatic and chemical interactions between a novel API and hundreds of potential binders, fillers, and lubricants, instantly flagging combinations that might lead to degradation over a two-year shelf life.
Digital Twins for Scale-Up
By creating a ‘digital twin’ of a continuous manufacturing line, engineers can simulate how an API will behave under the intense shear forces of a twin-screw extruder or the thermodynamics of a spray dryer. This allows for the precise optimisation of manufacturing parameters in silico, drastically reducing the amount of precious API wasted during physical scale-up trials.
Conclusion
The New Centre of Gravity
The narrative that biological discovery is the sole driver of pharmaceutical innovation is rapidly fading. As the molecules we design become increasingly complex, insoluble, and targeted, the burden of clinical success has shifted heavily onto Chemistry, Manufacturing, and Controls.
Today, an elegant molecule is merely a starting point. It is the sophisticated formulation science, the transition to continuous, sensor-driven manufacturing, and the deployment of predictive biostatistics that ultimately bridge the chasm between a laboratory curiosity and a life-saving human therapeutic.
In the modern era of drug development, mastering the CMC bottleneck is not just an operational requirement — it is the ultimate competitive advantage.
This article is intended for informational purposes and reflects the scientific and strategic perspectives of Dhee Lifesciences. It does not constitute regulatory or clinical advice. All proprietary methodologies referenced, including SynapTx, are the intellectual property of Dhee Lifesciences.