Camel Antibody Proteins:
A New Frontier in Biologics
How a chance discovery in camelid immunology is reshaping drug design, delivery, and the future of precision medicine.
Origins, Structure, and Why They Matter
The discovery that challenged immunology — and the structural elegance that followed.
The Discovery That Challenged Immunology
The modern story of camel antibody proteins begins in the early 1990s, when researchers studying camelid immune systems made an unexpected observation: alongside conventional antibodies, animals such as camels, llamas, and alpacas produce a distinct class of antibodies composed solely of heavy chains.
This discovery fundamentally challenged long-standing immunological assumptions. In humans and most mammals, antibodies are structurally complex molecules made up of two heavy chains and two light chains. Camelids, by contrast, generate heavy-chain–only antibodies, lacking light chains entirely. The functional binding unit — now widely known as a nanobody — is a single-domain fragment (VHH) that retains full antigen-binding capability.
Conventional Antibody
Nanobody (VHH)
This reduction in size is not merely structural — it is functionally transformative, opening biological doors that remain firmly closed to traditional antibodies.
Structural Elegance: Why Smaller Is More Powerful
Nanobodies are not simply “smaller antibodies”; they represent a distinct biological architecture optimised by millions of years of evolution. Key structural characteristics collectively enable them to bind targets inaccessible to traditional monoclonal antibodies:
Single-Domain Format (VHH)
Eliminates the need for heavy–light chain pairing, dramatically simplifying both structure and manufacture.
Compact Size (~3–4 nm)
Approximately one-tenth of conventional antibodies — enabling access to confined, cryptic binding sites.
Extended Binding Loops
Long CDR regions allow penetration of enzyme active sites, receptor clefts, and aggregated proteins.
High Stability
Resistant to heat, pH variation, and denaturation — a practical advantage for formulation and storage.
From a drug discovery standpoint, nanobodies expand the “druggable space” — accessing targets that have long been considered unreachable by conventional biologics.
A New Class Between Small Molecules and Antibodies
One of the most compelling aspects of camel-derived nanobodies is their positioning as a hybrid therapeutic class:
Small Molecules
Cell-permeable and manufacturable — but often non-specific, with off-target effects limiting utility in complex diseases.
Monoclonal Antibodies
Highly specific — but large (~150 kDa), poorly tissue-penetrant, and expensive to manufacture at scale.
Nanobodies occupy a compelling intermediate space: retaining high specificity whilst offering superior tissue penetration and modular engineering capability. Researchers increasingly describe them as a therapeutic paradigm bridging biologics and small-molecule pharmacology.
The Blood–Brain Barrier Problem — and Why Camel Proteins Matter
One of the most persistent challenges in modern drug development is the blood–brain barrier (BBB) — a highly selective physiological barrier that restricts most therapeutics from entering the brain. Traditional antibodies are simply too large (~150 kDa), exhibit poor penetration, and require high doses or invasive delivery.
Nanobodies, by contrast, are small enough to diffuse more effectively through biological barriers, and may cross into brain tissue via mechanisms inaccessible to larger proteins. Emerging studies suggest they can bind targets such as amyloid-beta and tau proteins — hallmarks of Alzheimer’s disease — with greater efficiency in preclinical models.
- Neurodegenerative diseases — Alzheimer’s and Parkinson’s disease
- CNS-targeted biologics with improved tissue penetration
- Reduced off-target effects due to higher binding specificity
- Potential applications in psychiatric and inflammatory CNS disorders
Early-Stage Evidence: Promise Without Overstatement
Whilst enthusiasm around camel antibody proteins is growing, scientific clarity is essential. The field is promising — but remains early-stage and requires rigorous human validation.
What Current Research Shows
- Nanobodies bind disease-relevant targets with high affinity
- Improved tissue penetration vs conventional antibodies
- Functional effects demonstrated in neurological disease models
- Potential behavioural improvements in experimental settings
What Remains Unresolved
- Long-term safety in human populations
- Pharmacokinetics — rapid renal clearance is a known limitation
- Optimal delivery strategies (IV, subcutaneous, intranasal)
- Large-scale clinical validation in neurodegeneration
From Curiosity to Platform Technology
What began as a biological curiosity has evolved into a platform technology with broad implications — spanning therapeutics, diagnostics, and drug delivery. Nanobodies are easier to engineer, faster to produce, and more adaptable to novel formats than conventional antibodies, enabling modular construction of multi-target constructs, combined delivery systems, and next-generation biologic platforms.
“Nanobodies are not simply smaller antibodies — they are a programmable molecular platform, where precision, adaptability, and delivery converge.”
Engineering, Translation, and the Road to Therapeutic Reality
From discovery to design — the science, challenges, and strategic opportunity of nanobody development.
From Discovery to Design: Engineering Nanobodies
Whilst camel-derived nanobodies originate in nature, their transformation into viable therapeutics is fundamentally an engineering process — relying on iterative design, selection, and optimisation to convert a naturally occurring binding domain into a clinically relevant molecule.
At the heart of this process lies phage display — a technique enabling the creation of vast libraries, often containing billions of variants, each displaying a slightly different nanobody sequence. These libraries are screened against a chosen target (such as amyloid-beta, inflammatory cytokines, or receptor proteins) to isolate candidates with high binding affinity, target specificity, and favourable biophysical properties. Lead candidates then undergo affinity maturation, where targeted mutations improve performance further.
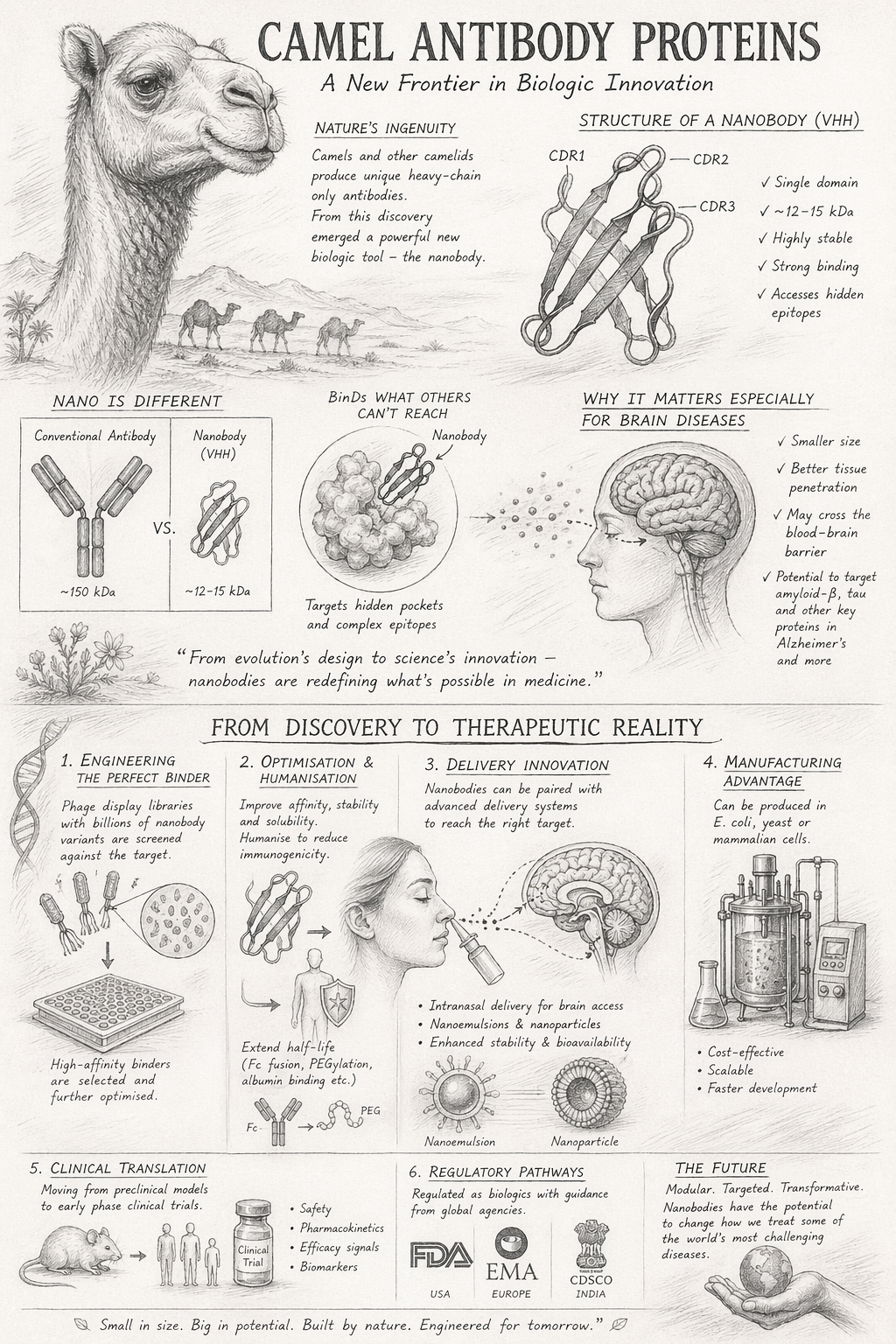
Camel-derived nanobodies (VHH domains) — structurally simplified yet functionally sophisticated single-domain antibody fragments derived from heavy-chain–only camelid antibodies.
Humanisation and Optimisation: Making Them Clinically Viable
Although nanobodies are derived from camelid proteins, their use in humans requires careful modification to minimise immunogenicity. Through antibody humanisation, specific amino acid residues are altered to resemble human antibody frameworks whilst preserving binding functionality. The balance is delicate: excessive modification risks loss of activity; insufficient modification may trigger immune responses.
In parallel, developers address one of the key limitations of nanobodies — their short systemic half-life. Due to small size, nanobodies are rapidly cleared via renal filtration. Three principal strategies are employed to overcome this:
- Fc domain fusion — to mimic the extended circulation of full antibodies
- PEGylation — increasing molecular size to slow renal clearance
- Albumin-binding domains — exploiting the long half-life of serum albumin
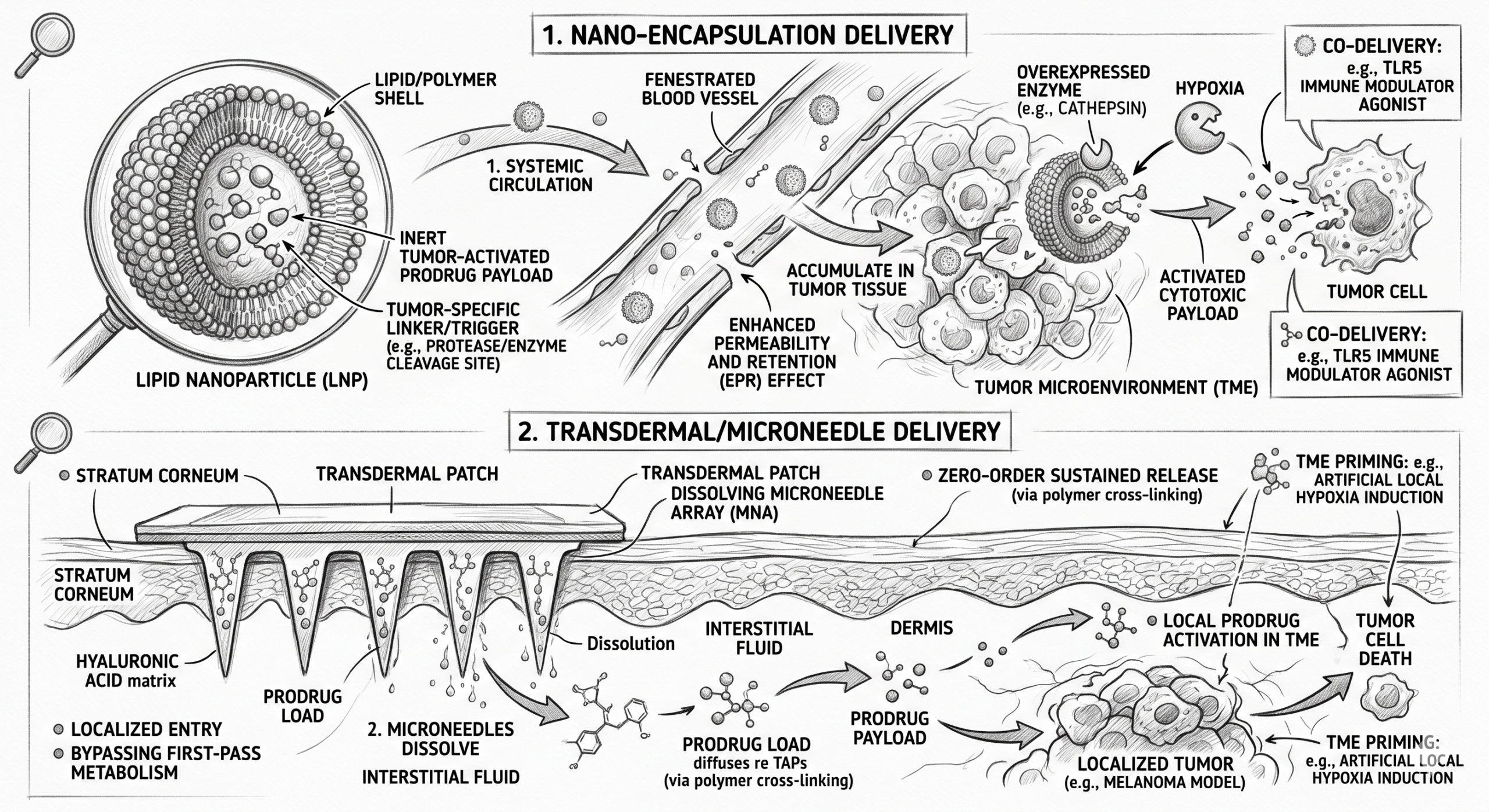
Delivery: The Defining Challenge and Opportunity
If nanobodies represent a breakthrough in targeting, delivery remains the decisive factor in their therapeutic success. The field is evolving rapidly — and significant innovation remains untapped.
Systemic Delivery
IV and subcutaneous routes remain the most established — but rapid clearance necessitates frequent dosing, and tissue distribution may remain suboptimal.
CNS-Focused Delivery
Smaller size improves tissue diffusion and potential BBB penetration. Emerging strategies include intranasal delivery, receptor-mediated transport, and carrier conjugation.
Advanced Formulations
Integration with nanoemulsions, lipid nanoparticles, and lyophilised systems may enhance bioavailability, enable targeting, and improve shelf-life at scale.
The convergence of nanobody biologics with advanced formulation science is likely to define the next wave of therapeutic innovation in this field — particularly for CNS and mucosal applications.
Manufacturing: A Quiet but Powerful Advantage
Unlike conventional monoclonal antibodies, nanobodies offer simplified and cost-efficient manufacturing pathways. They can be produced in Escherichia coli systems (low cost, high yield), yeast expression systems, or mammalian cells when complex modifications are required. This flexibility enables faster scale-up, lower production costs, and greater accessibility for emerging biotech ecosystems.
For markets such as India, this represents a genuine strategic advantage — allowing local development without reliance on high-cost biologics infrastructure.
Clinical Translation: Where Promise Meets Reality
Despite strong scientific rationale, the transition from laboratory success to clinical efficacy remains the most challenging step. Several nanobody-based products have entered clinical development across oncology, inflammatory diseases, and infectious diseases — whilst CNS indications, including Alzheimer’s disease, remain largely early-stage.
Key Clinical Challenges
- Demonstrating meaningful clinical endpoints in human trials
- Ensuring long-term safety across diverse patient populations
- Achieving consistent pharmacokinetics at scale
- Robust human data in neurodegeneration remains limited — preclinical promise must translate
Regulatory Pathways: Navigating a Hybrid Modality
Nanobodies occupy a regulatory position that is both familiar and novel — classified as biologics, yet structurally distinct from monoclonal antibodies. Regulatory oversight is guided by established frameworks (the FDA, EMA, and India’s CDSCO), but specific considerations apply:
- Immunogenicity assessment for engineered camelid-derived domains
- Validation of novel delivery routes, particularly intranasal administration
- Stability and formulation data for non-traditional dosage forms
For developers, early regulatory engagement is essential to avoid costly delays later in the development pathway.
Strategic Outlook: Platform, Not Product
Perhaps the most important shift in thinking is this: nanobodies are not just individual drug candidates — they are a platform technology. Their true value lies in modularity, compatibility with delivery systems, and rapid engineering cycles.
Direct development as novel biologic drugs across oncology, neurology, and inflammatory disease.
Conjugated to drug delivery systems (nanoparticles, liposomes) for precision payload delivery.
High specificity and small size make nanobodies ideal for molecular imaging and biosensor platforms.
Integration with small molecules or conventional biologics to create multi-modal therapeutic systems.
“For emerging companies in innovation-driven environments, the opportunity lies not merely in discovering a molecule — but in building a differentiated platform around it.”
From Molecule to Modality
Camel antibody proteins have progressed from an immunological curiosity to a serious contender in next-generation biologics. Yet their future will not be defined solely by binding properties — but by how effectively they are engineered, delivered, manufactured, and integrated into broader therapeutic strategies. The field remains early, but its direction is clear: nanobodies represent a shift from static biologics to programmable molecular systems, where precision, adaptability, and delivery converge.
This article is intended for informational purposes and reflects the scientific perspectives of Dhee Lifesciences. It does not constitute regulatory or clinical advice. The content references published preclinical research and emerging pharmacological literature.