-
Engineering molecules for development readiness
We integrate formulation science, predictive biopharmaceutics, and regulatory strategy to de-risk development and accelerate clinical success.
-
Predict clinical outcomes before development begins
Using predictive biopharmaceutics and simulation to anticipate human exposure, reduce uncertainty, and guide formulation and clinical strategy.
-
Translating science into clinical strategy
Integrating formulation design, pharmacokinetics, and clinical considerations into structured development pathways aligned for human studies.
-
Aligning development with regulatory execution
Designing CMC strategies and documentation frameworks to ensure seamless transition from development to clinical and manufacturing stages.
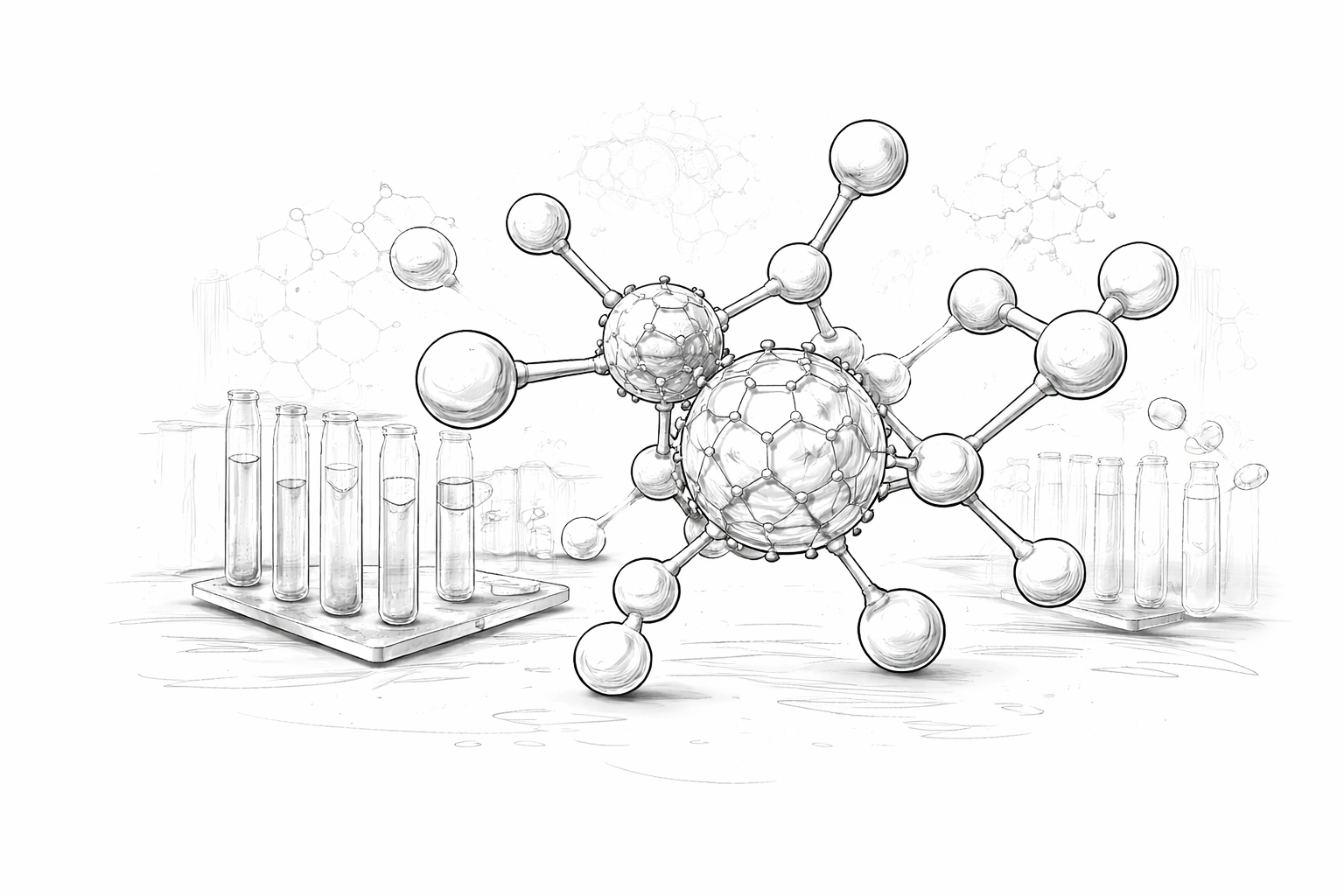



From molecule to clinic engineered, not assumed
We integrate predictive modelling, formulation engineering, and regulatory strategy into a single decision-driven pathway—reducing uncertainty, compressing timelines, and increasing clinical success probability.

Formulations engineered for predictable clinical exposure
Designed to overcome solubility, stability, and bioavailability challenges from the outset.

Predict before you commit
Human-relevant pharmacokinetic modelling to guide formulation and clinical strategy early.
Regulatory-aligned development from inception
Integrated CMC strategies designed to meet global regulatory expectations.

Built for seamless scale-up and transfer
Execution-ready documentation enabling efficient manufacturing and tech transfer.
Where we intervene in development
We partner at critical inflection points where molecules fail, stall, or require strategic redesign.
- Poor solubility or bioavailability limiting exposure
- Uncertain human pharmacokinetics and dose prediction
- Complex formulations requiring novel delivery systems
- Preclinical assets not translating into clinical success
- Molecules requiring rescue, repositioning, or lifecycle extension
- CMC and regulatory misalignment delaying progress
How we de-risk development —before failure occurs
Our approach integrates predictive modelling, formulation science, and regulatory strategy into a unified, decision-driven development system.
We simulate human pharmacokinetics and formulation behaviour early, reducing reliance on late-stage trial-and-error.
Every formulation strategy is informed by modelled outcomes — not empirical iteration alone.
We design development as a unified system — aligning formulation, clinical strategy, and regulatory expectations from the outset.
Development decisions are guided by clinical success criteria — not just formulation feasibility.
We ensure early development choices are compatible with manufacturing and tech transfer requirements.
Our proprietary platform integrates datasets, predictive models, and simulation tools to guide development decisions.
Integrated capabilities
We integrate molecular engineering, formulation science, predictive biopharmaceutics, and translational strategy to enable clinical exposure for complex, high-risk molecules.
—DEVELOPMENT REALITY
Drug development does not fail at discovery—it fails at translation.
The critical challenge lies in achieving predictable human exposure, stable formulation, and executable development pathways.
EXPOSURE
Human exposure is the real endpoint
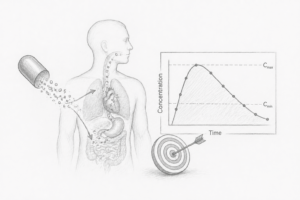
Most molecules fail because pharmacokinetic behaviour is not understood early—leading to unpredictable clinical outcomes.
FORMULATION
Formulation defines success

Solubility, stability, and delivery architecture determine whether a molecule can translate into a viable therapy.
CMC
Manufacturability is not an afterthought
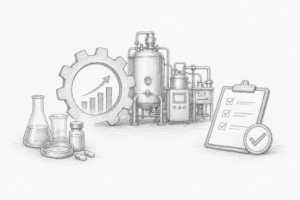
Development strategies must align with scalability, reproducibility, and regulatory expectations from the outset.
INTEGRATION
Fragmented development creates failure

Separating modelling, formulation, and clinical strategy leads to inefficiency. Integration is essential for execution.
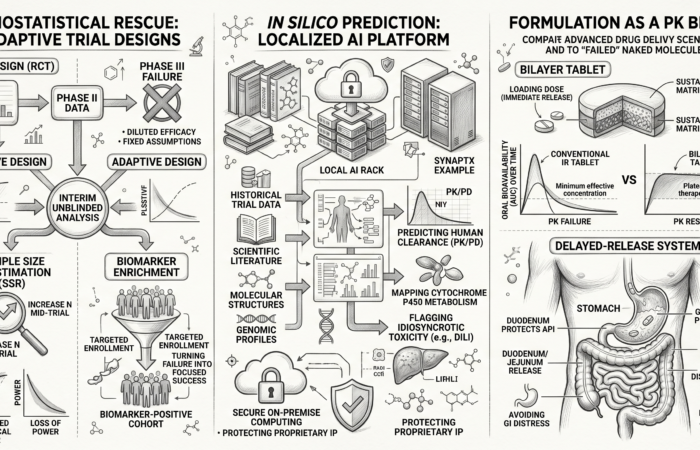
SynapTx™ — Our predictive development engine
A proprietary platform integrating data, predictive modelling, and simulation to guide formulation and clinical strategy before development begins.
01 01
Input
Molecular properties, preclinical data, and formulation variables are consolidated to define exposure, stability, and translational risk.
These inputs establish the foundational parameters that determine how a molecule behaves across biological and formulation environments.
02 02
Engine
Predictive modelling, and simulation algorithms analyse these inputs to predict human pharmacokinetics and system behaviour.
Decision frameworks evaluate multiple development pathways to identify strategies that are both clinically viable and manufacturable.
03 03
Output
Optimised formulation strategies and predicted human PK profiles are generated to guide development decisions.
These outputs translate into execution-ready pathways aligned with clinical, regulatory, and manufacturing requirements.
Selected development insights
Improving oral exposure
Bridging preclinical to clinical gap
Predict • Design • Develop • Translate • Scale
Development. Branding. Strategy.
Molecule Engineering. Predictive Modelling. Translational Science.
Data-driven decisions • Human-relevant modelling • Integrated development • Clinical readiness • Scalable execution
Proven across complex development programmes
Our work spans multiple therapeutic areas—but follows a single, repeatable framework.
Selected programmes demonstrating our ability to translate complex molecules into clinically and commercially viable assets.
- All
- Delivery & Exposure Engineering
- Molecule Rescue Platforms
- Next-Gen Translational Systems
Across diverse therapeutic areas and delivery challenges, our approach consistently enables translation from molecule to clinic.
Collaboration models
Strategic Development Partnerships
End-to-end co-development from molecule to clinical readiness
Targeted Problem-Solving Engagements
Focused interventions for formulation, PK, or translational challenges
Platform-Enabled Co-Development
Leveraging SynapTx™ for shared-risk, high-value programmes
We collaborate on molecules, biological questions, and enabling technologies to build clinically translatable assets.
Regulatory & Compliance







Insights in drug development & predictive biopharmaceutics
Perspectives on drug development, formulation science, and predictive biopharmaceutics.
De-risk your development pathway
Whether you're addressing formulation challenges, exploring predictive modelling, or preparing for clinical translation—we help you make better decisions, earlier.