
EXPERTISE
Predictive Biopharmaceutics
Using integrated biopharmaceutics and pharmacokinetic modelling to guide formulation, dosing, and development decisions with confidence.
Where We Deliver Impact

First-in-Human Readiness
Designing exposure-driven strategies that reduce uncertainty and improve early clinical success
Formulation Decision Support
Guiding formulation selection based on predicted in vivo performance
Exposure Optimisation
Ensuring therapeutic levels are achieved consistently across patient populations
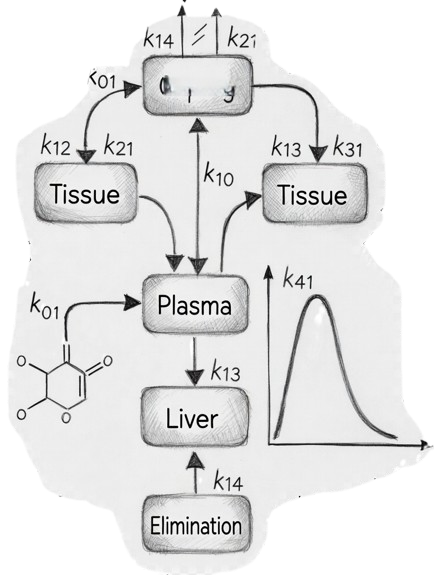
Mechanistic PK Modelling
Building models that describe absorption, distribution, metabolism, and elimination to predict human pharmacokinetics
Formulation–PK Alignment
Using modelling insights to guide formulation design toward achieving target exposure profiles
Biopharmaceutics Integration
Linking solubility, dissolution, and permeability characteristics to in vivo exposure outcomes
First-in-Human Strategy Design
Defining dose selection, escalation strategy, and exposure targets to reduce uncertainty in early clinical studies
Outcomes
- Reduced uncertainty in first-in-human studies
- Improved confidence in dose selection and escalation strategies
- Alignment of formulation performance with clinical exposure targets
- Reduced development timelines through informed decision-making
- Increased probability of clinical success through predictive insights



Where this fits in development
Applied across preclinical and early clinical stages to reduce translational risk, optimise first-in-human strategy, and enable data-driven development decisions.
Powered by SynapTx™
SynapTx™ integrates scientific literature, patent landscapes, and historical development data to enhance predictive modelling—enabling faster, evidence-driven decisions and identification of non-obvious risks and opportunities.
Looking to reduce uncertainty in your development program?
Partner with Dhee Lifesciences to design strategies that predict clinical exposure, optimise dosing, and accelerate confident decision-making.